When I compare a primary battery to a secondary battery, I see the most important difference is reusability. I use a primary battery once, then dispose of it. A secondary battery allows me to recharge and use it again. This impacts performance, cost, and environmental effects.
In summary, primary batteries offer single-use convenience, but secondary batteries support multiple uses and sustainability.
Key Takeaways
- Primary batteries provide reliable, single-use power with long shelf life, ideal for low-drain or emergency devices.
- Secondary batteries recharge hundreds to thousands of times, saving money and reducing waste in frequently used electronics.
- Choosing the right battery depends on device needs, balancing cost, convenience, and environmental impact for best results.
Primary Battery: Definition and Core Features

What Is a Primary Battery?
When I talk about a primary battery, I refer to a type of battery that stores energy for one-time use. After I use up the stored energy, I cannot recharge it. I find these batteries in many everyday items because they offer convenience and reliability.
In summary, a primary battery is a single-use power source that I cannot recharge.
How Primary Batteries Work
I see that a primary battery generates electricity through a chemical reaction inside the cell. The reaction happens only once. As I use the battery, the chemicals change and cannot return to their original state. This process makes the battery non-rechargeable.
To sum up, a primary battery works by converting chemical energy into electrical energy through a one-way reaction.
Common Types and Real-World Examples
I often use several types of primary batteries. The most common ones include:
- Alkaline batteries (used in remote controls and toys)
- Lithium primary batteries (found in cameras and smoke detectors)
- Coin cell batteries (used in watches and key fobs)
These batteries power devices that need steady, reliable energy for a limited time.
In short, I rely on primary batteries for devices that need dependable, single-use power.
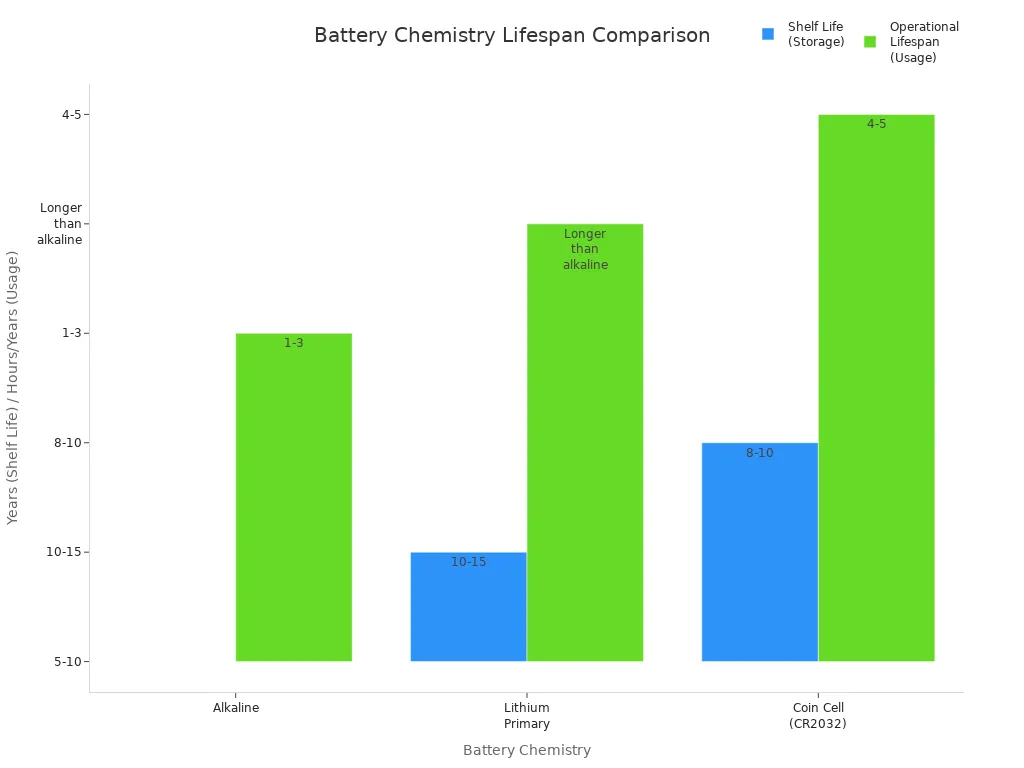
Usage and Lifespan Data
I always consider how long a primary battery lasts. Shelf life tells me how long the battery can sit unused and still work. Operational lifespan shows how long it powers a device. The table below helps me compare popular types:
| Battery Chemistry | Average Shelf Life (Storage) | Typical Operational Lifespan (Usage) | Key Notes on Usage and Longevity |
|---|---|---|---|
| Alkaline | 5-10 years | Varies; e.g., 1-3 hours in high-drain devices like digital cameras | Shelf life guaranteed up to 10 years by premium brands; zinc and manganese dioxide chemistry |
| Lithium Primary | 10-15 years | Longer operational lifespan due to low self-discharge; stable from -40°F to 122°F | Lithium metal chemistry offers superior stability and performance in extreme conditions |
| Coin Cell (e.g., CR2032) | 8-10 years | 4-5 years in key fobs; ~1 year in continuous-use devices like Apple AirTag | Ideal for low-drain, long-term applications |
I notice that environmental factors like temperature and humidity can shorten battery life. For best results, I store batteries at room temperature and moderate humidity.
In conclusion, primary batteries offer long shelf life and reliable performance, but actual usage time depends on the device and storage conditions.
Secondary Battery: Definition and Core Features

What Is a Secondary Battery?
When I discuss secondary batteries, I refer to electrochemical cells that I can recharge and use many times. Industry standards recognize these batteries as sustainable and cost-effective energy storage solutions. Unlike primary batteries, I do not throw them away after one use. I simply recharge them and continue using them for various applications.
In summary, a secondary battery is a rechargeable power source designed for repeated use.
How Secondary Batteries Work
I see that secondary batteries operate through reversible chemical reactions. When I charge the battery, electrical energy restores the original chemical state inside the cell. During use, the battery releases stored energy by reversing this process. This cycle repeats hundreds or even thousands of times, depending on the battery type and how I use it.
To sum up, secondary batteries work by allowing chemical reactions to go both ways, making recharging possible.
Common Types and Real-World Examples
I often encounter several types of secondary batteries in daily life:
- Nickel-metal hydride (Ni-MH) batteries: I use these in cordless phones and digital cameras.
- Lithium-ion (Li-ion) batteries: I find these in smartphones, laptops, and electric vehicles.
- Nickel-cadmium (Ni-Cd) batteries: I see these in power tools and emergency lighting.
These batteries power devices that need frequent charging and long-term reliability.
In short, secondary batteries are essential for modern electronics that require repeated energy cycles.
Usage and Lifespan Data
I always consider how long a secondary battery lasts. The table below shows typical cycle life and usage data for popular types:
| Battery Chemistry | Typical Cycle Life | Common Applications | Notes on Longevity |
|---|---|---|---|
| Ni-MH | 500–1,000 cycles | Cameras, toys, cordless phones | Good for moderate-drain devices |
| Li-ion | 300–2,000 cycles | Phones, laptops, EVs | High energy density, long life |
| Ni-Cd | 500–1,500 cycles | Power tools, emergency lights | Robust, tolerates deep discharge |
I notice that proper charging and storage extend battery life. High temperatures and overcharging can reduce performance.
In conclusion, secondary batteries offer long-term value through multiple charge cycles and reliable performance when I use them correctly.
Key Differences Between Primary and Secondary Battery
Reusability and Rechargeability
When I compare these two battery types, I see a clear difference in how I use them. I use a primary battery once, then replace it when it runs out. I cannot recharge it. In contrast, I recharge a secondary battery many times. This feature makes secondary batteries ideal for devices I use every day, like smartphones and laptops. I find that reusability not only saves me money over time but also reduces waste.
In summary, I use a primary battery for single-use applications, while I rely on secondary batteries for repeated use and recharging.
Chemical Reactions and Energy Storage
I notice that the chemical reactions inside these batteries work differently. In a primary battery, the chemical reaction moves in one direction. Once the chemicals react, I cannot reverse the process. This makes the battery non-rechargeable. With a secondary battery, the chemical reaction is reversible. When I charge the battery, I restore the original chemical state, allowing me to use it again.
Recent advancements have improved both types:
- Lithium-ion batteries now reach energy densities up to 300 Wh/kg.
- Solid-state electrolytes make batteries safer and more efficient.
- Silicon-based anodes and new cell designs push energy densities even higher.
- Researchers are exploring sodium-ion and metal-air batteries for future applications.
To sum up, I see that primary batteries use one-way chemical reactions, while secondary batteries use reversible reactions that enable recharging and higher energy storage.
Lifespan and Performance Data
I always consider how long a battery lasts and how well it performs. A primary battery usually has a long shelf life, sometimes up to 10 years, but I can only use it once. Its operational lifespan depends on the device and usage. Secondary batteries offer hundreds or even thousands of charge cycles. For example, lithium-ion batteries can last from 300 to over 2,000 cycles, especially with new technologies targeting even longer life for electric vehicles and grid storage.
| Battery Type | Shelf Life (Storage) | Cycle Life (Recharge) | Typical Use Case |
|---|---|---|---|
| Primary battery | 5–15 years | 1 (single-use) | Remote controls, clocks |
| Secondary battery | 2–10 years | 300–5,000+ cycles | Phones, laptops, EVs |
In conclusion, I choose a primary battery for long shelf life and single use, but I select a secondary battery for repeated use and longer overall lifespan.
Cost Comparison with Real-World Figures
When I look at costs, I see that a primary battery often costs less upfront. For example, a pack of four AA alkaline batteries might cost $3–$5. However, I need to replace them after each use. A secondary battery, such as a rechargeable AA Ni-MH cell, may cost $2–$4 each, but I can recharge it up to 1,000 times. Over time, I spend less money by choosing rechargeable batteries for high-use devices.
In summary, I pay more initially for secondary batteries, but I save money in the long run if I use them frequently.
Environmental Impact and Recycling Statistics
I recognize that battery choice affects the environment. When I use a primary battery, I create more waste because I dispose of it after one use. Secondary batteries help reduce waste because I recharge and reuse them. However, both types present recycling challenges. Recycling rates for batteries remain low worldwide, and resource scarcity is a growing concern. New battery chemistries, such as solid-state and sodium-ion, aim to use more sustainable materials and improve recycling efficiency.
To sum up, I help the environment by choosing secondary batteries for frequent use and by recycling all batteries properly whenever possible.
Advantages and Disadvantages of Primary Battery
Benefits with Supporting Data
When I choose a primary battery, I see several clear advantages. I notice that these batteries offer long shelf life, which means I can store them for years without losing much power. I rely on primary batteries for devices that need instant, reliable energy, such as flashlights and medical equipment. I find that primary batteries perform well in low-drain devices, like remote controls and wall clocks. I appreciate the convenience because I do not need to recharge them. I can use them straight out of the package.
Here are some key benefits:
- Long shelf life: Alkaline primary batteries can last up to 10 years in storage.
- Immediate usability: I do not need to charge before use.
- Wide availability: I can buy primary batteries almost anywhere.
- Stable performance: These batteries deliver consistent voltage until depleted.
Tip: I always keep a pack of primary batteries for emergencies because they work reliably even after years in storage.
Advantages and Disadvantages of Secondary Battery
Benefits with Supporting Data
When I use secondary batteries, I see many advantages that make them a smart choice for modern devices. I can recharge these batteries hundreds or even thousands of times, which saves me money over the long term. I notice that lithium-ion batteries, for example, can last up to 2,000 cycles if I use and charge them properly. This means I do not need to buy new batteries as often.
I also find that secondary batteries help reduce waste. By reusing the same battery, I throw away fewer batteries each year. According to the U.S. Environmental Protection Agency, rechargeable batteries can cut household battery waste by up to 80%. I see that these batteries work well in high-drain devices like smartphones, laptops, and power tools.
Key benefits I experience:
- Long-term cost savings due to reusability
- Lower environmental impact
- High performance in demanding devices
- Consistent voltage output during use
In summary, I choose secondary batteries for their cost-effectiveness, strong performance, and positive impact on the environment.
Drawbacks with Supporting Data
I also recognize some challenges when I use secondary batteries. I pay more upfront for rechargeable batteries compared to single-use ones. For example, a lithium-ion battery can cost two to three times more than an alkaline battery. I must also use a charger, which adds to my initial investment.
Secondary batteries can lose capacity over time. After hundreds of charge cycles, I notice that the battery holds less energy. For instance, a typical Ni-MH battery may drop to 80% of its original capacity after 500 cycles. I also need to handle and store these batteries carefully to avoid damage or safety risks.
| Drawback | Example/Supporting Data |
|---|---|
| Higher initial cost | Li-ion: $5–$10 vs. Alkaline: $1–$2 |
| Capacity loss over time | Ni-MH: ~80% capacity after 500 cycles |
| Requires charger | Extra purchase needed |
To sum up, I weigh the higher upfront cost and gradual capacity loss against the long-term savings and convenience of secondary batteries.
Choosing the Right Battery Type
Best Uses for Primary Battery
I reach for a primary battery when I need instant power in devices that do not require frequent replacement. I use these batteries in emergency flashlights, wall clocks, and remote controls. I notice that medical devices, such as hearing aids and glucose meters, often rely on primary batteries because they deliver stable voltage and long shelf life. I prefer primary batteries for backup situations since they hold charge for years and work right out of the package.
Key point: I choose a primary battery for devices that need reliable, single-use energy and long-term storage.
Best Uses for Secondary Battery
I select secondary batteries for electronics that demand regular charging and high performance. I use rechargeable batteries in smartphones, laptops, and cameras. I rely on secondary batteries for power tools and electric vehicles because they support hundreds or thousands of charge cycles. I find these batteries ideal for toys, wireless headphones, and game controllers, where frequent use makes recharging practical and cost-effective.
Key point: I use secondary batteries for devices that require repeated charging and consistent power over time.
Real-World Examples and Statistics
I see clear trends in battery usage across industries. According to market data, over 80% of households use primary batteries in remote controls and smoke detectors. I notice that rechargeable batteries now power more than 90% of smartphones and laptops worldwide. In the automotive sector, electric vehicles rely exclusively on secondary batteries, with lithium-ion cells supporting up to 2,000 charge cycles. I observe that switching from disposable to rechargeable batteries can reduce household battery waste by up to 80%.
| Device Type | Preferred Battery Type | Typical Usage Frequency | Notable Statistic |
|---|---|---|---|
| Remote control | Primary battery | Occasional | 80% of homes use disposables |
| Smartphone | Secondary battery | Daily | 90%+ use rechargeable batteries |
| Electric vehicle | Secondary battery | Continuous | 2,000+ charge cycles possible |
Key point: I match battery type to device needs, using primary batteries for low-drain, infrequent use and secondary batteries for high-drain, frequent use.
I choose a primary battery for low-drain devices that I use infrequently. I rely on secondary batteries for electronics that demand frequent charging. I always consider cost, convenience, and environmental impact before making a decision. The right battery type helps me save money and reduce waste.
Key point: I match battery choice to device needs for best results.
FAQ
What devices work best with primary batteries?
I use primary batteries in low-drain devices like remote controls, wall clocks, and emergency flashlights.
Key point: I choose primary batteries for devices that need reliable, single-use power.
How many times can I recharge a secondary battery?
I recharge secondary batteries hundreds or thousands of times, depending on the chemistry and usage.
| Battery Type | Typical Recharge Cycles |
|---|---|
| Ni-MH | 500–1,000 |
| Li-ion | 300–2,000 |
Key point: I select secondary batteries for frequent charging and long-term use.
Are rechargeable batteries better for the environment?
I reduce battery waste by using rechargeable batteries. I help lower landfill impact and conserve resources.
- Rechargeable batteries cut household battery waste by up to 80%.
Key point: I support sustainability by choosing rechargeable batteries whenever possible.
Post time: Aug-22-2025




